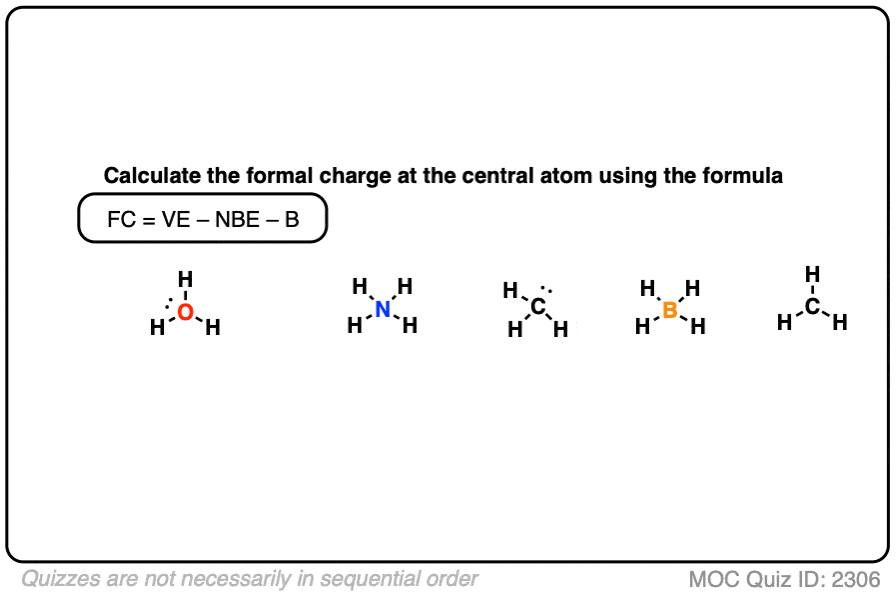  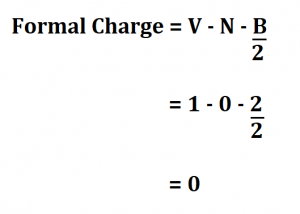
Formal charge of valence electrons electrons in lone pairs + 1/2 the number of bonding electrons Since the number of bonding electrons divided by 2 is equal to the number of bonds surrounding the atom, this formula can be shortened to: Formal Charge of valence electrons on atom non-bonded electrons. The equation to determine the Formal Charge on an atom: FC (The number of valence electrons on the atom in the ground state) ½ (total number of electrons in covalent bonds attached to the atom, with each covalent bond having two electrons) (all electrons in lone pairs on an atom, with each lone pair equal to two electrons). Yes, just switch the single and double bonds for the above two structures (with the oxygens and their lone electrons). The formula for calculating the formal charge on an atom is simple. Hybridization, Lewis Structure, & Formal Charge 2. Calculating Formal Charge: The following equation determines the formal charge for each atom in a molecule or polyatomic ion. The difference between the atoms number of valence electrons and the number it owns is the formal charge. Image: Single Replacement Compounds that have the same chemical formula but different structural. In the left resonance structure, all the atoms have zero formal charge, while on the right structure, the nitrogen has a +1 formal charge, and the oxygen with the single bond has a -1 formal charge.Ĭan you draw two additional resonance structures for the above molecules? Count all of its lone pair electrons, and half of its bonding electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
